Multiphoton microscopy Triple the fluorescence signal
In multi photon microscopy, adaptive optics are implemented on the excitation pathway and can significantly boost the fluorescence signal from a biological sample. Correction of the system and sample-induced aberrations improves the focusing ability of the microscope, with a typical gain of 3 to 5 times the two-photon effect. Starting by determining the optical aberrations and using a deformable mirror to correct them. The increase in fluorescence signal can either allow much deeper layers of the sample to be reached or decreasing the intensity of the excitation laser in order to prevent sample bleaching and minimize photo-toxicity effects.
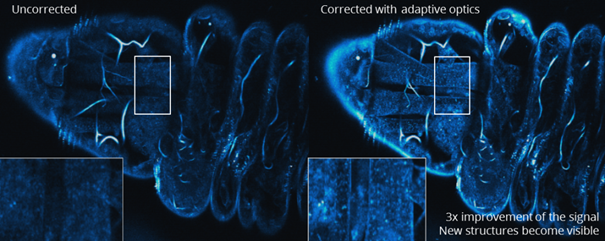
Figure caption: Drosophila larva imaging by Third Harmonic Generation microscopy (THG) without (left) and with (right) adaptive optics. Correction of aberrations in this case boosts the fluorescence signal by a factor of 3, and new features inside the sample become visible (see zooms). Image produced in collaboration with Emmanuel Beaurepaire, LOB, Ecole Polytechnique, France.